Parabens have been a hot topic for years, and they’ve been accused of all evils. While it is important to point out that they haven’t exactly been proven innocent in every case, we must also recognise that parabens don’t necessarily all deserve the bad rap they get in the media. That is why we’ve decided to examine the question in detail.
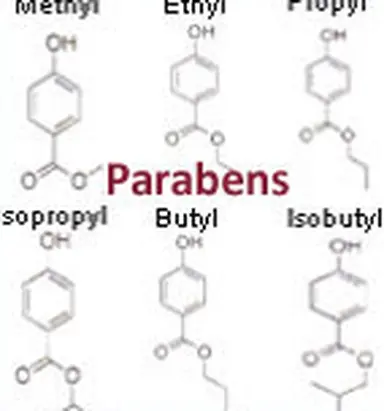
Parabens actually make up a family of ingredients. They all resemble each other rather closely, but their chemical structures differ (particularly the length of their alkyl chains), as do their scopes of action and their potential adverse effects. Clearly stated, there is not just one paraben but many, which can be used alone or in combination, to improve their action and increase efficacy.
Up until 2014, six parabens were primarily used in cosmetics. Here they are, in increasing order by alkyl chain length, as they appear on labels in the official list of cosmetic ingredients: Methylparaben, Ethylparaben, Propylparaben, Isopropylparaben, Butylparaben, Isobutylparaben.
Recognised efficacy
Parabens are preservatives (parahydroxybenzoic acid esters), and they are very widely used to protect cosmetic products from bacterial and microbiological proliferation. Due to their broad scope of activity on bacteria, moulds, yeasts, and fungi, as well as their complementary nature when acting synergistically (when more than one are mixed together), they were crucial, must-have ingredients for several years. It is estimated that at their peak (about two or three years ago), they preserved nearly 80% of hygiene and beauty products, all categories combined, all around the world. For formulators and many observers, they were considered to be …