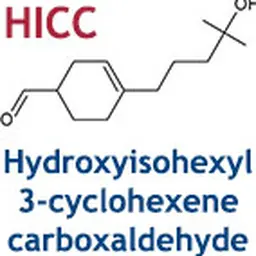
The European Union has just transmitted to the WTO a draft Regulation aiming to ban or regulate substances classified as CMR by Regulation 2021/849 (17th ATP to CLP), published in May 2021. On the agenda: 14 new entries in Annex II (prohibited substances) to the Cosmetics Regulation 1223/2009. The text also foresees the inclusion in Annex III (restricted substances) of Methyl Salicylate (fragrance ingredient) and corrections in Annexes II and V for the entries concerning Sodium Hydroxymethylglycinate (preservative).
Notification Date
3 Febuary 2022
Products covered
Cosmetics
Title of the notified document
Draft Commission Regulation amending Regulation (EC) No.1223/2009 of the European Parliament and of the Council as regards the use in cosmetic products of certain substances classified as carcinogenic, mutagenic or toxic for reproduction and correcting that Regulation.
Content
The draft measure is required to enact the prohibition to use as cosmetic ingredients substances classified as carcinogenic, mutagenic or toxic for reproduction (CMR) by Commission Regulation (EU) No.2021/849, which has been adopted based on the CLP Regulation and will apply from 17 December 2022. The adoption of this draft Regulation is, therefore, needed to reflect in the Cosmetics Regulation the new CMRs classification provided by Commission Regulation (EU) No.2021/849. In addition, in order to ensure legal certainty and a high level of protection of human health, entry 1669 of Annex II to Regulation (EC) No.1223/2009 has been deleted and entry 51 of Annex V has been corrected to reflect the wording used in notes 8 and 9 to the CMR classification and the correct chemical name of the substance Sodium N-(hydroxymethyl)glycinate.
Objective
Protection of human health or safety
The text of the future Regulation
Whereas
Regulation (EC) No.1272/2008 …