A peptide is a linear chain of amino acids (AA); a protein is also a linear chain of amino acids, but it is a much longer chain. The boundary between the two is not officially fixed, but from a molar mass of 10,000 daltons (approx. 100 amino acids), we speak more of proteins than peptides. An oligopeptide (which we will focus on more specifically in the following) is composed of 2 to about 20 AA, followed by polypeptides.
History of peptides in cosmetics: Part 1 of a series
A peptide is a linear chain of amino acids (AA); a protein is also a linear chain of amino acids, but it is a much longer chain. The boundary between the two is not officially fixed, but from a molar mass of 10 000 daltons (approx. 100 amino acids), we speak more of proteins than of peptides. An oligopeptide (the entity in which we will focus more precisely in what follows) is composed of 2 to about 20 AA, after which we speak of polypeptides.
Like Mr. Jourdan and his prose in Molière's"Bourgeois Gentilhomme", many cosmetic formulators have been using peptides in their creams or shampoos consciously or unconsciously for a long time. So why is there so much talk of"peptides" in the current communication on the sophisticated"active ingredients" of today's products?
When a beautiful protein obtained on a large scale by extraction from a natural source (collagen from the skin, casein from milk, keratin from the hair or feathers of birds, wheat, soya, potato, lupin proteins…) is taken and subjected to a gentle hydrolysis (either enzymatically or with strong acid), a mixture of peptide fragments is obtained, the size composition of which (i.e. chain length, AA distribution…) varies from one protein to another, or even from one production to another, since hydrolysis is not very specific and the result is somewhat random. It's like cutting a large ball of multicoloured wool with scissors at random through the whole ball. The many pieces of yarn recovered will be of different length and colour, and, from batch to batch, different in detail, although statistically the piles of small pieces would roughly resemble each other.
Therefore, products with the INCI name"wheat/soya/milk/milk protein hydrolysate","hydrolyzed collagen/elastin/keratin/silk…" all composed of a heap of"peptides", were already found in cosmetic products of all kinds for a very long time..: inexpensive preparations, easy to use, bringing a little touch, a little hydration through hygroscopy, a little sheathing on the hair, even cuticle strengthening, these hydrolysates were (and still are) useful and popular basic ingredients, even if marketing gurus rarely put them forward as representatives of innovation.
Except that… with the introduction of specific bioactive peptides (see below) and the recognition of their growing interest, their triumphal march equaling that of the AHAs of the 1990s or Retinol of the 1990s, we rediscovered that these protein hydrolysates were also"peptides". Admittedly, the composition was not well known analytically, variable from batch to batch, and the biological activity of these mixed fragments was only very rarely studied. But we could say that the night cream, the shampoo, the foundation contained"peptides"… Not wrong, but not very informative either.
It was at the beginning of the 1990s, following the BSE ("mad cow") crisis which"cleaned" the field of active ingredients of all"animal contamination", that synthetic peptides, of high biological specificity, made their first appearance.
A collagen fragment, composed of three amino acids (Gly-His-Lys = GHK) complexed with copper ions Cu 2+ "("copper petide") had been identified by Maquart et al. as being able to stimulate collagen synthesis in fibroblasts, a useful activity in connective tissue healing. The only problem was that a peptide of this nature (loaded with two H+ and one O-) and such solubility in water had practically no chance of penetrating through the stratum corneum to reach the skin fibroblasts. Copper, a metal considered useful and necessary for the construction of the extracellular matrix, eventually played a beneficial role, without the question"Gly-His-Lys? or Copper? or both? being necessary for action?
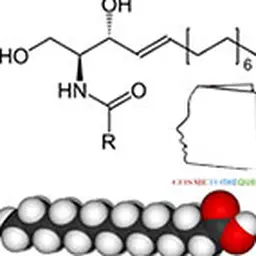
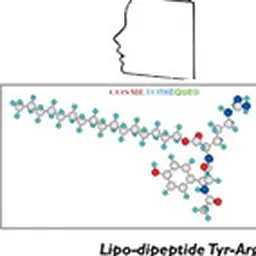
The Sederma's work were able to bypass the obstacle: by grafting a fatty acid (C 16 = palmitoyl) on the peptide, trans-epidermal penetration was assured and activity in vitro of this lipopeptide (stimulation of collagen synthesis) was increased tenfold compared to the non-modified peptide. So much so that a tiny amount (3-5 ppm) of this lipopeptide in an anti-aging formula could give a measurable anti-wrinkle effect in clinical studies, even compared to the peptide-free formula, often referred to as the"anti-wrinkle" formula. placebo" Note: the notion of"placebo" has no meaning in cosmetics since it is the integral product that will act on the skin (or hair) with all its components. To test the activity of a single ingredient, one should rather speak of a"control product" or"vehicle"]. Given the very high cost of a custom peptide synthesized on a small industrial scale, and the reluctance of researchers and formulators to believe that such a small quantity of an"active" could generate undeniable benefits for the skin, coupled with the difficulty for Marketing to communicate on this unprecedented new concept, the start of synthetic bioactive peptides was very slow.
A hexapeptide (fragment of the elastin VGVAPG) also provided with a palmitoyl chain, a stimulator of elastic tissue, followed GHK and thus other peptides, all natural fragments of proteins present in the human body, prepared the ground before the great breakthrough in the year 2000.
Before talking about it (2 e and following parts), here are some general technical details to know about the use of these peptides in cosmetics:
- peptides are generally used in very small quantities, in the ppm range, - they are presented to formulators in the form of ready-to-use solutions, - the sequence (sequence of the sequence of AA in the chain) is very important: a GHK peptide (collagen synthesis) will not have the same activity as a GKH peptide (lipolysis), - small peptides (<10 AA) have no or very little allergenic risk and also very little toxicological risk in general : due to their low concentration in the products, a few nano grams per cm are applied topically 2 of skin, From this, a small amount penetrates to the dermis or into the blood, where proteolytic enzymes metabolize them in minutes, peptides are generally chosen to be identical or analogous to protein fragments already present in the body ( Matrikin principle ) which further reduces the risk of side effects, - a difficulty in the use of peptides is linked to this low dosage: how to detect analytically some 3 or 5 ppm of peptide in the >50 ingredients that make up a skin care cream today? The LC/MS or MS/MS Maldi/TOF methods can solve the problem in some cases.
This was the situation of peptides in cosmetics before 2000. Continued in the second part of this story .
Karl Lintner
It did take us a little time to understand that putting more and more assets was not a solution and that this path proposed an interesting new paradigm. We were in a logic of"always more" at that time. Today, there is no doubt!
JCLJ
|
Karl is a Chemical Engineer from the Technical University of Vienna (Austria) and holds a PhD in Biochemistry from the same University. After 10 years of research on Biological Peptides at Centre d'Études Nucléaires de Saclay, he joined HENKEL KGaA in Düsseldorf (Germany). In 1990, he became Technical Director at SEDERMA (Active Cosmetic Ingredients Development), then General Manager. Among the many achievements of this company, the introduction of the Peptides concept to cosmetic applications ("MATRIXYL®") is particularly noteworthy. Winner of several Innovation Awards for SEDERMA, he is the recipient of the In-Cosmetics Life Time Achievement Award 2013. Associate professor at the UVSQ, he is an internationally recognized lecturer in the world of cosmetics. |

 This contribution was made by Karl Lintner.
This contribution was made by Karl Lintner.