Regulation 2017/1410 of 2 August 2017, published in Official Journal of the European Union on 3 August, amends Annexes II (prohibited substances) and III (restricted substances) of Cosmetics Regulation 1223/2009, to prohibit the use of three fragrance ingredients. This provision will apply from 23 August 2019 and will be fully implemented from 23 August 2021.
The whereas of Regulation 2017/1410
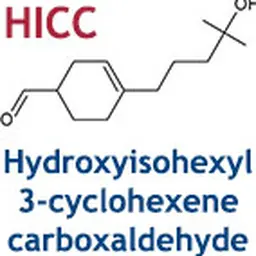
(1) The Scientific Committee on Consumer Safety (SCCS) concluded in its Opinion of 26-27 June 2012 that 3- and 4-(4-Hydroxy-4-methylpentyl) cyclohex-3-ene-1-carbaldehyde (HICC), with the INCI name of Hydroxyisohexyl 3-Cyclohexene Carboxaldehyde, 2,6-Dihydroxy-4-methyl-benzaldehyde (Atranol) and 3-Chloro-2,6-Dihydroxy-4-methyl-benzaldehyde (Chloroatranol) should not be used in cosmetic products as they are the fragrance allergens which caused the highest number of contact allergies cases in past years.
(2) Consequently, there is a potential risk to human health and those substances should therefore be prohibited in cosmetic products.
(3) HICC is regulated in entry 79 of Annex III to Regulation (EC) No.1223/2009, where it is provided that its presence must be indicated in the list of ingredients referred to in Article 19(1)(g) of that Regulation when its concentration exceeds 0.001% in leave-on products and 0.01% in rinse-off products. Since HICC should be prohibited in cosmetic products, it is necessary to delete that entry.
(4) The SCCS indicated in its opinion of 26-27 June 2012 that atranol and chloroatranol are natural components of oak tree moss (Evernia Prunastri) and treemoss (Evernia Furfuracea) extracts regulated in entry 91 and 92, respectively, of Annex III to Regulation (EC) No.1223/2009.
(5) It is appropriate to provide for …