It is usually considered friendly and festive as a drink (if it is consumed with moderation, obviously), but can alcohol keep the same image as a cosmetic ingredient? That remains to be seen… And even if it is used a lot, both the advantages and drawbacks of its presence in our hygiene and beauty products should be analyzed according to various data, especially if they are used on a daily basis.
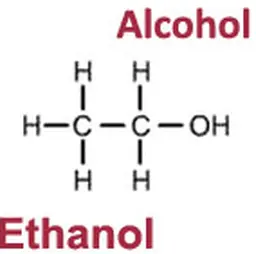
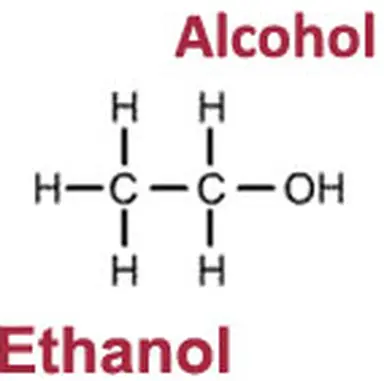
CH₃-CH₂-OH. Behind this chemical formula lies ethanol, or ethyl alcohol, commonly known as just ‘alcohol’.
It is a primary alcohol obtained either by fermenting plants rich in sugars (fruit, cereal, sugar cane…), or with distillation. As a result, it can be called ‘natural’, or even sustainable, since it is agro-sourced and obtained with ‘green’ processes (extraction, fermentation, and distillation for beetroot; extraction, hydrolysis, fermentation, and distillation for cereal like wheat or corn). It can even be derived from organic farming.
Colourless and volatile, it is obviously contained in alcoholic drinks (wine, beer, aperitifs, liqueurs…), but also in a great number of cosmetics, as a raw material with multiple applications… On top of that, it is cheap.
Alcohol galore!
Of course, it is mostly an essential in perfumery, as it makes up 70 to 95% of perfume bottles. It solubilizes the perfumed concentrate, which is composed of lipophilic components soluble in alcohol. It is also transparent, easily sprayed, and, due to its volatility, it evaporates after the application, so that only the perfumed concentrate is left on the skin.
Alcohol as a cosmetic active
It can make up from 0 to 20% of skincare formulas and fulfil various functions:
• Refreshing …